If you think diabetes treatment is just about controlling blood sugar — the latest guidelines say otherwise.
The American Diabetes Association (ADA) released major updates to its Standards of Care in both 2025 and 2026, and the core message is clear: type 2 diabetes treatment must now protect your heart, kidneys, and weight — not just your HbA1c number.
Here’s what changed:
From one drug at a time → to combination therapy from the start
The 2025 guidelines moved away from the old “try one drug, wait, then add another” approach. Doctors are now advised to start combination therapy early — especially when a patient already has heart disease, kidney disease, or obesity.
Newer drugs promoted to front-line treatment
GLP-1 receptor agonists (like semaglutide) and SGLT2 inhibitors (like empagliflozin) are no longer reserved as “add-on” drugs. For high-risk patients, these medications should be prescribed based on their organ-protective benefits — regardless of current blood sugar levels or metformin use.
Weight and liver health now part of the conversation
The 2025–2026 guidelines also formally include fatty liver disease (MASLD) and obesity as conditions that should shape treatment decisions in type 2 diabetes.
Your diabetes doctor is no longer just watching your blood sugar — they’re now protecting your heart, kidneys, and weight all at once
Reference: ADA Standards of Care in Diabetes — 2025 Official Release | Exploration Publishing — 2025 ADA Guideline Analysis
Tirzepatide (Mounjaro) — dual GLP-1/GIP results
Most diabetes medications work on one hormone pathway. Tirzepatide works on two — and that difference shows up dramatically in the results.
Sold as Mounjaro for type 2 diabetes treatment, tirzepatide is the first drug to activate both the GLP-1 and GIP receptors simultaneously. Think of these as two natural gut hormones that tell your body to release insulin, reduce appetite, and burn fat. Activating both together produces results that neither hormone achieves alone.
What the clinical trials show:
- HbA1c (3-month blood sugar average) reduced by 1.9% to 2.6% across SURPASS Phase 3 trials -among the highest reductions ever seen in a diabetes drug
- Average body weight loss of 15–22% at 72 weeks, depending on dose
- In the 3-year SURMOUNT-1 trial, tirzepatide reduced the risk of progressing from pre-diabetes to type 2 diabetes by 94% compared to placebo
- In September 2025, the SURPASS-PEDS trial published in The Lancet showed it reduced HbA1c by 2.2% in children and teens (ages 10–17) with type 2 diabetes
Head-to-head trials also show tirzepatide outperforms semaglutide (Ozempic/Wegovy) on both blood sugar control and weight loss.
Tirzepatide isn’t just a diabetes drug — in a 3-year trial, it reduced the risk of developing type 2 diabetes by 94% in people with pre-diabetes.
Results vary by individual. Side effects include nausea and GI discomfort. Consult your doctor before starting.
References: Eli Lilly — SURPASS-PEDS Trial Results (2025) | Eli Lilly — SURMOUNT-1 Three-Year Data | PMC — Tirzepatide Systematic Review & Meta-Analysis
CGM Is Now Recommended for Non-Insulin Type 2 Diabetes — A Game-Changer
Until 2025, continuous glucose monitors (CGMs) were largely seen as tools for people on insulin. That’s no longer the case.
What is a CGM?
A CGM is a small sensor — usually worn on the arm or abdomen — that measures your blood sugar every few minutes, automatically, all day and night. No finger pricks needed. It sends readings to your phone in real time.
What changed in 2025?
The ADA’s 2025 guidelines formally recommend considering CGM use for adults with type 2 diabetes who are on non-insulin glucose-lowering medications — like metformin, SGLT2 inhibitors, or GLP-1 drugs. This is the first time this population has been explicitly included.
The 2026 update went even further, recommending CGM use from the time of diagnosis for anyone who could benefit — removing earlier restrictions tied to treatment type or HbA1c levels.
Why does this matter for patients?
- Real-time glucose data helps you understand exactly how your meals, exercise, and stress affect blood sugar
- Studies show CGM use reduces HbA1c even in people not on insulin
- It helps doctors fine-tune your treatment faster, without waiting months for a lab report
You don’t need to be on insulin to benefit from a CGM anymore — your glucose sensor can now be your most powerful diabetes management tool.
📚 References: The American Diabetes Association Releases Standards of Care in Diabetes—2025 | American Diabetes Association
GLP-1 Agonists for Heart and Kidney Protection – Beyond Blood Sugar
Here’s something most diabetes patients don’t know: some of the newest diabetes medications are now being prescribed primarily to protect your heart and kidneys not just to lower blood sugar.
GLP-1 receptor agonists (like semaglutide, sold as Ozempic) were originally developed as glucose lowering drugs. But large clinical trials have revealed something far more significant — they actively protect vital organs.
What the evidence shows:
The landmark FLOW trial — the first dedicated kidney outcomes trial for a GLP-1 drug — showed that semaglutide reduced:
- Major kidney events by 24%
- Major cardiovascular events (heart attacks, strokes) by 18%
- Death from any cause by 20%
compared to placebo, in patients with type 2 diabetes and chronic kidney disease (CKD).
Based on these results, in January 2025, the FDA expanded semaglutide’s approved uses to specifically include reducing the risk of kidney disease progression and cardiovascular death in adults with type 2 diabetes and CKD.
A broader meta-analysis of 11 trials covering over 85,000 patients confirmed that GLP-1 drugs as a class reduce kidney failure risk by 16% and all-cause death by 12%.
The key takeaway: If you have type 2 diabetes and also have heart disease, heart failure, or kidney disease, the 2025 ADA guidelines say your doctor should consider a GLP-1 drug — regardless of your current blood sugar level.
Semaglutide isn’t just lowering blood sugar anymore — it’s now FDA-approved to protect your kidneys and reduce the risk of cardiovascular death in diabetic kidney disease.
These medications require a prescription and medical supervision. Benefits and risks vary by individual health profile.
📚 References: FLOW Trial — NEJM Full Publication |
FDA Semaglutide Expanded Indication — PMC NIH |
Lancet Meta-Analysis — GLP-1 Kidney & CV Outcomes
Gut Microbiome and Type 2 Diabetes — The Emerging Frontier
Your gut is home to trillions of bacteria. And science is now discovering that the health of those bacteria is directly linked to whether you develop — or can better manage — type 2 diabetes.
What is the gut microbiome?
It’s the entire community of microorganisms living in your digestive tract. When this community is balanced, it supports healthy metabolism. When it’s disrupted- a state called dysbiosis — things can go wrong.
How does it connect to diabetes?
Research shows that people with type 2 diabetes consistently have a different gut bacteria profile compared to non-diabetic individuals. Specifically:
- A reduction in butyrate-producing bacteria — these are the “good” bacteria that reduce gut inflammation and improve insulin sensitivity
- An increase in harmful bacteria that raise systemic inflammation, directly worsening insulin resistance
- Disrupted gut barrier function, allowing toxins to leak into the bloodstream and trigger further metabolic damage
Interestingly, certain gut bacteria also influence how much GLP-1 your body naturally produces — the same hormone that diabetes drugs like semaglutide mimic.
What can be done now?
Research on probiotics, prebiotics, high-fibre diets, and even faecal microbiota transplantation (FMT) shows promising effects on blood sugar control. However, most studies are still in early stages — this is not yet a standalone treatment.
Practical steps backed by evidence: eat more fibre, fermented foods (like curd/yoghurt), and diverse vegetables. Avoid unnecessary antibiotic use.
The bacteria in your gut may be silently worsening your insulin resistance — and fixing them could become the next big frontier in diabetes treatment.
Gut microbiome therapies are still emerging. Do not replace prescribed diabetes medications with supplements without consulting your doctor.
References: Nutrients Journal — Gut Microbiome & T2DM Strategies (2025) |
PMC — Gut Microbiota Dysbiosis & T2DM Pathogenesis (2025) |
PMC — Gut Microbiota & Diabetes Treatment Frontiers
AI-Powered Diabetes Management Tools — Your Phone as a Health Partner
Artificial intelligence is no longer just a tech buzzword. In diabetes care, it’s already changing how patients monitor, predict, and manage their blood sugar every single day.
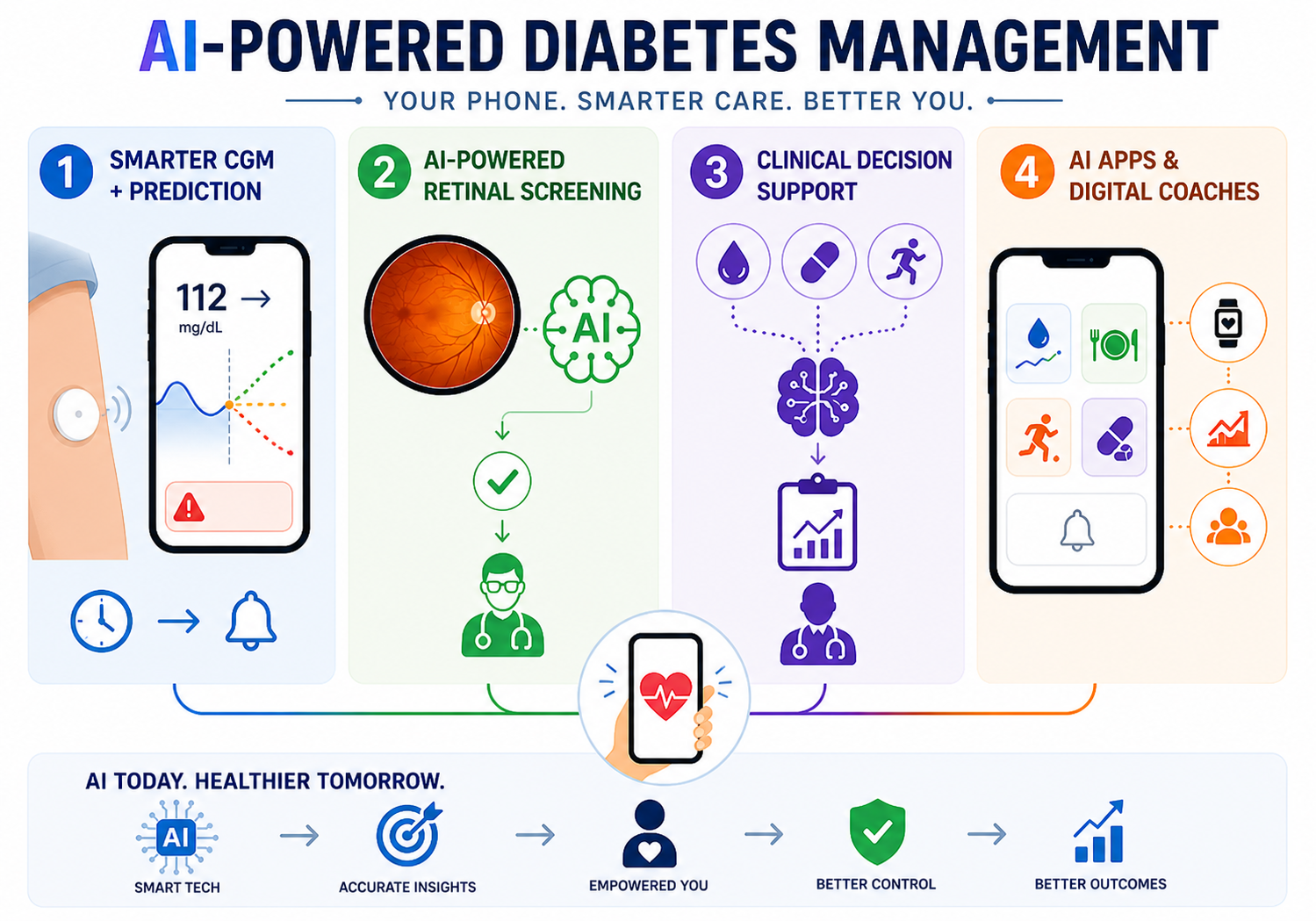
Here’s where AI is making a real difference right now:
1. Smarter CGM + Glucose Prediction
AI algorithms integrated with CGM sensors can now predict blood sugar levels up to 60 minutes in advance — alerting patients before a dangerous high or low actually happens. This is a leap beyond traditional CGMs that only show you where your glucose is right now.
2. AI-Powered Retinal Screening
Diabetic retinopathy (eye damage caused by diabetes) is a leading cause of blindness. AI systems can now analyse retinal photographs and detect early-stage retinopathy with accuracy comparable to specialist doctors — making screening faster and more accessible, especially in under-served areas.
3. Clinical Decision Support
AI tools are helping doctors analyse patterns across a patient’s glucose data, medications, and lifestyle — providing personalised treatment recommendations that would take a human clinician much longer to derive.
4. Diabetes Apps & Digital Coaches
Mobile apps now combine CGM data, meal logging, physical activity, and medication reminders into one AI-driven dashboard, helping patients make smarter real-time decisions without waiting for their next doctor’s appointment.
AI can now predict your blood sugar crash before it happens — diabetes management in 2025 is smarter than ever.
AI tools assist diabetes management — they do not replace your doctor or prescribed medications. Always use them under medical supervision
📚 References: PMC — AI in Diabetes Care: Comprehensive Review |
Springer — AI & Digital Health in Diabetes (2025) |
PMC — AI-Driven Innovations in Diabetes Care
What’s in the Pipeline — Stem Cells, Gene Therapy & the Future of Type 2 Diabetes
The treatments we’ve covered so far are available today. But what’s coming next could be even more transformative — potentially addressing the root cause of diabetes rather than just managing its symptoms.
Stem Cell Therapy
Scientists are developing ways to grow healthy, insulin-producing beta cells from stem cells in a laboratory and transplant them into patients. Early clinical trials show improved blood sugar control, reduced insulin requirements, and in some cases, insulin independence. Companies like Vertex Pharmaceuticals are already in Phase 1/2 human trials for stem cell-derived beta cell therapies.
CRISPR Gene Editing
In a landmark 2025 development, researchers successfully transplanted CRISPR-edited pancreatic islet cells that produced insulin for months — without requiring immunosuppressive drugs. CRISPR technology is also advancing for monogenic forms of diabetes (where a single gene mutation is the cause), with the goal of a one-time treatment that corrects the problem at its source.
What this means for type 2 diabetes specifically
While much of the early work targets type 1 diabetes, gene therapy trials for correcting insulin-resistance mutations and regenerating beta cell function in type 2 diabetes are underway and progressing.
Important reality check: These therapies are not yet available to most patients. Most trials are in Phase 1–2 stages. Be cautious of unverified “stem cell clinics” offering unproven treatments — always seek care from a certified endocrinologist.
📚 References: European Society of Medicine — Stem Cell Therapy in T2DM (2025) |
ScienceDirect — Gene & Stem Cell Therapy Future in Diabetes (2025) |
Health.gheware — CRISPR Beta Cell Transplant 2025 Breakthrough
Conclusion:
The landscape of type 2 diabetes treatment is evolving faster than ever, offering more hope and better outcomes for patients today than in any previous decade. Whether you are newly diagnosed or looking to optimize your current regimen, the goal remains the same: stable blood sugar and a high quality of life. Don’t view your treatment as a set of restrictions, but as a roadmap to wellness. Take it one day at a time, stay consistent with your check-ups, and remember that every small healthy choice adds up to a major impact on your future health.
FAQ:
What happens if you get diagnosed with type 2 diabetes?
A type 2 diabetes diagnosis means your body struggles to regulate blood sugar. Management involves setting a target A1c, starting medications like Metformin, and adopting a diet rich in complex carbohydrates. You’ll likely begin routine glucose monitoring and regular screenings for eye and foot health. While chronic, it is highly manageable through a combination of clinical care, physical activity, and consistent lifestyle adjustments.
Best medicine for type 2 diabetes?
There is no single “best” medicine; treatment is individualized. Metformin is the standard first-line choice for its safety and efficacy. For those with heart or kidney concerns, GLP-1 agonists (like Ozempic) or SGLT2 inhibitors (like Jardiance) are often preferred for their protective benefits and weight loss potential. The optimal choice depends on a patient’s specific health profile and must be determined by a healthcare provider.
What level of blood sugar is dangerous for type 2 diabetes?
Dangerous levels typically include hypoglycemia (below 70 mg/dL), which requires immediate sugar intake, and severe hyperglycemia (above 250–300 mg/dL). Extremely high readings can lead to life-threatening conditions like Hyperosmolar Hyperglycemic State
First line treatment for type 2 diabetes?
Metformin is the globally recognized first-line pharmacological treatment for type 2 diabetes. It is preferred due to its long-term safety record, effectiveness in lowering HbA1c, low cost, and minimal risk of causing hypoglycemia.
About the author:
Mohammad Junaid Rain is a 2nd-year MBBS student focused on simplifying diabetes care for Indian families. He creates evidence-based health content using medical textbooks, ADA guidance, ICMR resources, research papers, and clinical exposure. His goal is to make diabetes management practical and easy to understand for Indians.
Disclaimer: This article is for educational purposes only and does not substitute professional medical advice. Please consult your doctor for diagnosis and treatment.

